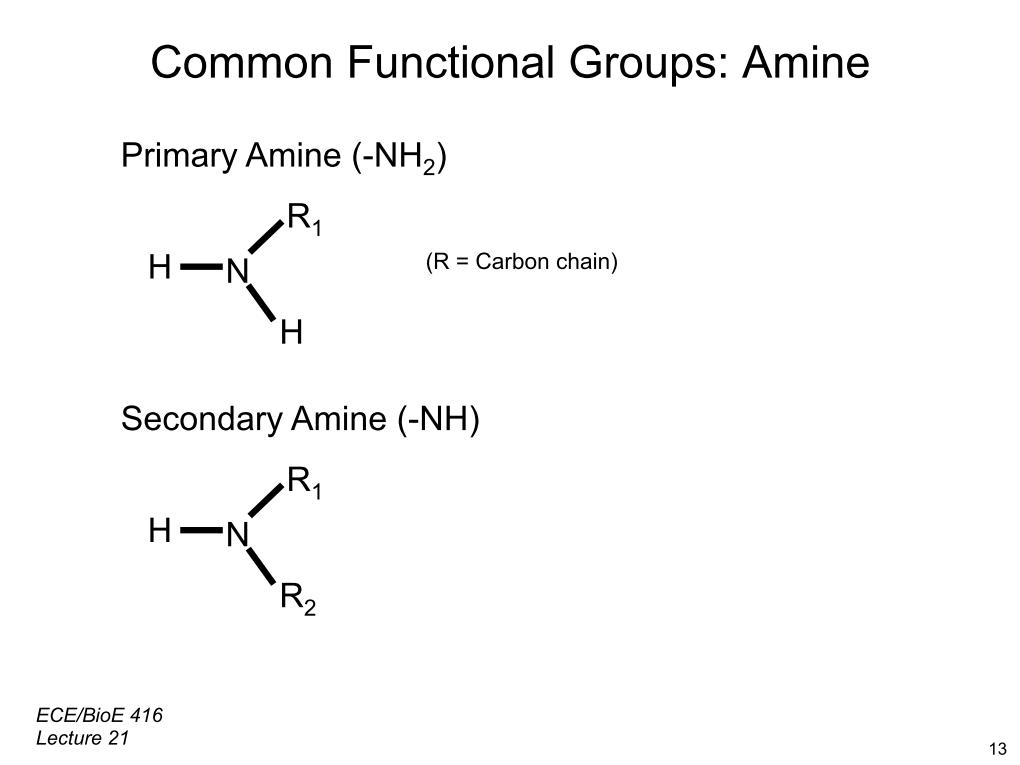
Their boiling points are high and their solubility in water lowĪmines of the type NHRR' and NRR'R" are chiral: the nitrogen atom bears four substituents counting the lone pair. The aromatic amines, such as aniline, have their lone pair electrons conjugated into the benzene ring, thus their tendency to engage in hydrogen bonding is diminished. Primary amines react with ketones such as acetone, and most amines are incompatible with chloroform and carbon tetrachloride. Aliphatic amines display significant solubility in organic solvents, especially polar organic solvents. Solubility decreases with the increase in the number of carbon atoms. Gaseous amines possess a characteristic ammonia smell, liquid amines have a distinctive "fishy" smell.Īlso reflecting their ability to form hydrogen bonds, most aliphatic amines display some solubility in water. Thus methylamine and ethylamine are gases under standard conditions, whereas the corresponding methyl alcohol and ethyl alcohols are liquids. Thus the boiling point of amines is higher than those of the corresponding phosphines, but generally lower than those of the corresponding alcohols. Hydrogen bonding significantly influences the properties of primary and secondary amines. Higher amines have the prefix amino as a functional group.Ģ-aminopentane (or sometimes: pent-2-yl-amine or pentane-2-amine) Systematic names for some common amines: Lower amines are named with the suffix -amine. An organic compound with multiple amino groups is called a diamine, triamine, tetraamine and so forth. Typically, the compound is given the prefix "amino-" or the suffix: "-amine." The prefix "N-" shows substitution on the nitrogen atom. Naming conventionsĪmines are named in several ways. The presence of an amine group strongly increases the reactivity of the aromatic ring, due to an electron-donating effect. The aromatic ring decreases the alkalinity of the amine, depending on its substituents. Aromatic aminesĪromatic amines have the nitrogen atom connected to an aromatic ring as in anilines. These compounds are not amines but are called quaternary ammonium cations, have a charged nitrogen center, and necessarily come with an anion. It is also possible to have four alkyl substituents on the nitrogen. N-methylpiperidine is a cyclic tertiary amine. Examples of cyclic amines include the 3-member ring Aziridine and the six-membered ring piperidine. Cyclic amines are either secondary or tertiary amines. Examples include trimethylamine, a distinctively fishy smell. Important representatives include dimethylamine and methylethanolamine.In tertiary amines, all three hydrogen atoms are replaced by organic substituents. Secondary amines have two alkyl substituents bound to N together with one hydrogen. Important primary alkyl amines include methylamine, ethanolamine (2-aminoethanol), and the buffering agent tris). Primary amines arise when one of three hydrogen atoms in ammonia is replaced by an alkyl.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
